Ulcerative colitis
| Ulcerative colitis | |
|---|---|
| Classification and external resources | |
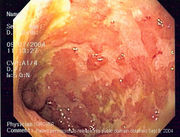
 Endoscopic image of a sigmoid colon afflicted with ulcerative colitis. Note the vascular pattern of the colon granularity and focal friability of the mucosa. |
|
| ICD-10 | K51. |
| ICD-9 | 556 |
| OMIM | 191390 |
| DiseasesDB | 13495 |
| MedlinePlus | 000250 |
| eMedicine | med/2336 |
| MeSH | D003093 |
Ulcerative colitis (Colitis ulcerosa, UC) is a form of inflammatory bowel disease (IBD). Ulcerative colitis is a form of colitis, a disease of the intestine, specifically the large intestine or colon, that includes characteristic ulcers, or open sores, in the colon. The main symptom of active disease is usually constant diarrhea mixed with blood, of gradual onset. Because of the name, IBD is often confused with irritable bowel syndrome ("IBS"), a troublesome, but much less serious, condition. Ulcerative colitis has similarities to Crohn's disease, another form of IBD. Ulcerative colitis is an intermittent disease, with periods of exacerbated symptoms, and periods that are relatively symptom-free. Although the symptoms of ulcerative colitis can sometimes diminish on their own, the disease usually requires treatment to go into remission.
Ulcerative colitis occurs in 35–100 people for every 100,000 in the United States,[1] or less than 0.1% of the population. The disease is more prevalent in northern countries of the world, as well as in northern areas of individual countries or other regions. Although ulcerative colitis has no known cause, there is a presumed genetic component to susceptibility. The disease may be triggered in a susceptible person by environmental factors. Although dietary modification may reduce the discomfort of a person with the disease, ulcerative colitis is not thought to be caused by dietary factors. Although ulcerative colitis is treated as though it were an autoimmune disease, there is no consensus that it is such. Treatment is with anti-inflammatory drugs, immunosuppression, and biological therapy targeting specific components of the immune response. Colectomy (partial or total removal of the large bowel through surgery) is occasionally necessary, and is considered to be a cure for the disease.
Contents |
Causes
While the cause of ulcerative colitis is still unknown, several, possibly interrelated, causes have been suggested.
Genetic factors
A genetic component to the etiology of ulcerative colitis can be hypothesized based on the following:[2]
- Aggregation of ulcerative colitis in families.
- Identical twin concordance rate of 10% and dizygotic twin concordance rate of 3%[3]
- Ethnic differences in incidence
- Genetic markers and linkages
There are 12 regions of the genome which may be linked to ulcerative colitis. This includes chromosomes 16, 12, 6, 14, 5, 19, 1, 16, and 3 in the order of their discovery.[4] However, none of these loci has been consistently shown to be at fault, suggesting that the disorder arises from the combination of multiple genes. For example, chromosome band 1p36 is one such region thought to be linked to inflammatory bowel disease.[5] Some of the putative regions encode transporter proteins such as OCTN1 and OCTN2. Other potential regions involve cell scaffolding proteins such as the MAGUK family. There are even HLA associations which may be at work. In fact, this linkage on chromosome 6 may be the most convincing and consistent of the genetic candidates.[4]
Multiple autoimmune disorders have been recorded with the neurovisceral and cutaneous genetic porphyrias including ulcerative colitis, Crohn's disease, celiac disease, dermatitis herpetiformis, diabetes, systemic and discoid lupus, rheumatoid arthritis, ankylosing spondylitis, scleroderma, Sjogren's disease and scleritis. Physicians should be on high alert for porphyrias in families with autoimmune disorders and care must be taken with potential porphyrinogenic drugs, including sulfasalazine.
Environmental factors
Many hypotheses have been raised for environmental contributants to the pathogenesis of ulcerative colitis. They include the following:
- Diet: as the colon is exposed to many dietary substances which may encourage inflammation, dietary factors have been hypothesized to play a role in the pathogenesis of both ulcerative colitis and Crohn's disease. There have been few studies to investigate such an association, but one study showed no association of refined sugar on the prevalence of ulcerative colitis.[6]
- Diet: A diet low in fermentable dietary fiber may affect ulcerative colitis incidence.
- Breastfeeding: There have been conflicting reports of the protection of breastfeeding in the development of inflammatory bowel disease. One Italian study showed a potential protective effect.[7]
- Several scientific studies have posted that Accutane is a possible trigger of Crohn's Disease and Ulcerative colitis in some individuals. Three cases in the United States have gone to trial thus far, with all three resulting in multi-million dollar judgements against the makers of isotretinoin. There were an additional 425 cases pending as of .[8][9][10][11][12]
Autoimmune disease
Some sources list ulcerative colitis as an autoimmune disease,[13] a disease in which the immune system malfunctions, attacking some part of the body. In contrast to Crohn's disease, which can affect areas of the gastrointestinal tract outside of the colon, ulcerative colitis usually involves the rectum and is confined to the colon, with occasional involvement of the ileum. This so-called "backwash ileitis" can occur in 10–20% of patients with pancolitis and is believed to be of little clinical significance.[14] Ulcerative colitis can also be associated with comorbidities that produce symptoms in many areas of the body outside the digestive system. Surgical removal of the large intestine often cures the disease.[15]
Alternative theories
Levels of sulfate-reducing bacteria tend to be higher in persons with ulcerative colitis. This could mean that there are higher levels of hydrogen sulfide in the intestine. An alternative theory suggests that the symptoms of the disease may be caused by toxic effects of the hydrogen sulfide on the cells lining the intestine.[16][17] It may be caused by occlusions in the capillaries of the subepithelial linings, degenerated fibers beneath the mucosa and infiltration of the lamina propria with plasma cells.
Epidemiology
The incidence of ulcerative colitis in North America is 10–12 cases per 100,000 per year, with a peak incidence of ulcerative colitis occurring between the ages of 15 and 25. Prevalence is 1 per 1000. There is thought to be a bimodal distribution in age of onset, with a second peak in incidence occurring in the 6th decade of life. The disease affects females more than males.[18]
The geographic distribution of ulcerative colitis and Crohn's disease is similar worldwide,[19] with highest incidences in the United States, Canada, the United Kingdom, and Scandinavia. Higher incidences are seen in northern locations compared to southern locations in Europe [20] and the United States.[21]
As with Crohn's disease, the prevalence of ulcerative colitis is greater among Ashkenazi Jews and decreases progressively in other persons of Jewish descent, non-Jewish Caucasians, Africans, Hispanics, and Asians.[14] Appendectomy prior to age 20 for appendicitis[22] and tobacco use[23] are protective against development of ulcerative colitis.
Clinical presentation
GI symptoms
The clinical presentation[18] of ulcerative colitis depends on the extent of the disease process. Patients usually present with diarrhea mixed with blood and mucus, of gradual onset. They also may have signs of weight loss, and blood on rectal examination. The disease is usually accompanied with different degrees of abdominal pain, from mild discomfort to severely painful cramps.
Ulcerative colitis is associated with a general inflammatory process that affects many parts of the body. Sometimes these associated extra-intestinal symptoms are the initial signs of the disease, such as painful, arthritic knees in a teenager. The presence of the disease cannot be confirmed, however, until the onset of intestinal manifestations.
Extent of involvement

Ulcerative colitis is normally continuous from the rectum up the colon. The disease is classified by the extent of involvement, depending on how far up the colon the disease extends:
- Distal colitis, potentially treatable with enemas:[15]
- Proctitis: Involvement limited to the rectum.
- Proctosigmoiditis: Involvement of the rectosigmoid colon, the portion of the colon adjacent to the rectum.
- Left-sided colitis: Involvement of the descending colon, which runs along the patient's left side, up to the splenic flexure and the beginning of the transverse colon.
- Extensive colitis, inflammation extending beyond the reach of enemas:
- Pancolitis: Involvement of the entire colon, extending from the rectum to the cecum, beyond which the small intestine begins.
Severity of disease

In addition to the extent of involvement, UC patients may also be characterized by the severity of their disease.[15]
- Mild disease correlates with fewer than four stools daily, with or without blood, no systemic signs of toxicity, and a normal erythrocyte sedimentation rate (ESR). There may be mild abdominal pain or cramping. Patients may believe they are constipated when in fact they are experiencing tenesmus, which is a constant feeling of the need to empty the bowel accompanied by involuntary straining efforts, pain, and cramping with little or no fecal output. Rectal pain is uncommon.
- Moderate disease correlates with more than four stools daily, but with minimal signs of toxicity. Patients may display anemia (not requiring transfusions), moderate abdominal pain, and low grade fever, 38 to 39 °C (100 to 102 °F).
- Severe disease, correlates with more than six bloody stools a day, and evidence of toxicity as demonstrated by fever, tachycardia, anemia or an elevated ESR.
- Fulminant disease correlates with more than ten bowel movements daily, continuous bleeding, toxicity, abdominal tenderness and distension, blood transfusion requirement and colonic dilation (expansion). Patients in this category may have inflammation extending beyond just the mucosal layer, causing impaired colonic motility and leading to toxic megacolon. If the serous membrane is involved, colonic perforation may ensue. Unless treated, fulminant disease will soon lead to death.
Extraintestinal features


As ulcerative colitis is believed to have a systemic (i.e., autoimmune) origin, patients may present with comorbidities leading to symptoms and complications outside the colon. These include the following:
- aphthous ulcers of the mouth
- Ophthalmic (involving the eyes):
- Iritis or uveitis, which is inflammation of the iris
- Episcleritis
- Musculoskeletal:
- Seronegative arthritis, which can be a large-joint oligoarthritis (affecting one or two joints), or may affect many small joints of the hands and feet
- Ankylosing spondylitis, arthritis of the spine
- Sacroiliitis, arthritis of the lower spine
- Cutaneous (related to the skin):
- Erythema nodosum, which is a panniculitis, or inflammation of subcutaneous tissue involving the lower extremities
- Pyoderma gangrenosum, which is a painful ulcerating lesion involving the skin
- Deep venous thrombosis and pulmonary embolism
- Autoimmune hemolytic anemia
- clubbing, a deformity of the ends of the fingers
- Primary sclerosing cholangitis, a distinct disease that causes inflammation of the bile ducts
Similar conditions
The following conditions may present in a similar manner as ulcerative colitis, and should be excluded:
- Crohn's disease
- Infectious colitis, which is typically detected on stool cultures
- Pseudomembranous colitis, or Clostridium difficile-associated colitis, bacterial upsets often seen following administration of antibiotics
- Ischemic colitis, inadequate blood supply to the intestine, which typically affects the elderly
- Radiation colitis in patients with previous pelvic radiotherapy
- Chemical colitis resulting from introduction of harsh chemicals into the colon from an enema or other procedure.
Comparison to Crohn's disease
The most common disease that mimics the symptoms of ulcerative colitis is Crohn's disease, as both are inflammatory bowel diseases that can affect the colon with similar symptoms. It is important to differentiate these diseases, since the course of the diseases and treatments may be different. In some cases, however, it may not be possible to tell the difference, in which case the disease is classified as indeterminate colitis.
| Crohn's Disease | Ulcerative Colitis | |
|---|---|---|
| Involves terminal ileum? | Commonly | Seldom |
| Involves colon? | Usually | Always |
| Involves rectum? | Seldom | Usually |
| Peri-anal involvement? | Commonly | Seldom |
| Bile duct involvement? | Not associated | Higher rate of Primary sclerosing cholangitis[24] |
| Distribution of Disease | Patchy areas of inflammation | Continuous area of inflammation |
| Endoscopy | Linear and serpiginous (snake-like) ulcers | Continuous ulcer |
| Depth of inflammation | May be transmural, deep into tissues | Shallow, mucosal |
| Fistulae, abnormal passageways between organs | Commonly | Seldom |
| Biopsy | May have non-necrotizing non-peri-intestinal crypt granulomas[25][26] | Crypt abscesses and cryptitis |
| Surgical cure? | Often returns following removal of affected part | Usually cured by removal of colon, can be followed by pouchitis |
| Smoking | Higher risk for smokers | Lower risk for smokers |
| Autoimmune disease? | Generally regarded as an autoimmune disease | No consensus |
| Cancer risk? | Lower than ulcerative colitis | Higher than Crohn's |
Diagnosis and workup
General
_active.jpg)
The initial diagnostic workup for ulcerative colitis includes the following:[1][15]
- A complete blood count is done to check for anemia; thrombocytosis, a high platelet count, is occasionally seen
- Electrolyte studies and renal function tests are done, as chronic diarrhea may be associated with hypokalemia, hypomagnesemia and pre-renal failure.
- Liver function tests are performed to screen for bile duct involvement: primary sclerosing cholangitis.
- X-ray
- Urinalysis
- Stool culture, to rule out parasites and infectious causes.
- Erythrocyte sedimentation rate can be measured, with an elevated sedimentation rate indicating that an inflammatory process is present.
- C-reactive protein can be measured, with an elevated level being another indication of inflammation.
Although ulcerative colitis is a disease of unknown causation, inquiry should be made as to unusual factors believed to trigger the disease.[15] Factors may include: recent cessation of tobacco smoking; recent administration of large doses of iron or vitamin B6; hydrogen peroxide in enemas or other procedures.
Endoscopic
_endoscopic_biopsy.jpg)
The best test for diagnosis of ulcerative colitis remains endoscopy. Full colonoscopy to the cecum and entry into the terminal ileum is attempted only if diagnosis of UC is unclear. Otherwise, a flexible sigmoidoscopy is sufficient to support the diagnosis. The physician may elect to limit the extent of the exam if severe colitis is encountered to minimize the risk of perforation of the colon. Endoscopic findings in ulcerative colitis include the following:
- Loss of the vascular appearance of the colon
- Erythema (or redness of the mucosa) and friability of the mucosa
- Superficial ulceration, which may be confluent, and
- Pseudopolyps.
Ulcerative colitis is usually continuous from the rectum, with the rectum almost universally being involved. There is rarely peri-anal disease, but cases have been reported. The degree of involvement endoscopically ranges from proctitis or inflammation of the rectum, to left sided colitis, to pancolitis, which is inflammation involving the ascending colon.
Histologic
Biopsies of the mucosa are taken to definitively diagnose UC and differentiate it from Crohn's disease, which is managed differently clinically. Microbiological samples are typically taken at the time of endoscopy. The pathology in ulcerative colitis typically involves distortion of crypt architecture, inflammation of crypts (cryptitis), frank crypt abscesses, and hemorrhage or inflammatory cells in the lamina propria. In cases where the clinical picture is unclear, the histomorphologic analysis often plays a pivotal role in determining the diagnosis and thus the management. By contrast, a biopsy analysis may be indeterminate, and thus the clinical progression of the disease must inform its treatment.
Course and complications
Progression or remission
Patients with ulcerative colitis usually have an intermittent course, with periods of disease inactivity alternating with "flares" of disease. Patients with proctitis or left-sided colitis usually have a more benign course: only 15% progress proximally with their disease, and up to 20% can have sustained remission in the absence of any therapy. Patients with more extensive disease are less likely to sustain remission, but the rate of remission is independent of the severity of disease.
Ulcerative colitis and colorectal cancer
There is a significantly increased risk of colorectal cancer in patients with ulcerative colitis after 10 years if involvement is beyond the splenic flexure. Those with only proctitis or rectosigmoiditis usually have no increased risk.[15] It is recommended that patients have screening colonoscopies with random biopsies to look for dysplasia after eight years of disease activity.[27]
Primary sclerosing cholangitis
Ulcerative colitis has a significant association with primary sclerosing cholangitis (PSC), a progressive inflammatory disorder of small and large bile ducts. As many as 5% of patients with ulcerative colitis may progress to develop primary sclerosing cholangitis.[28]
Mortality
The effect of ulcerative colitis on mortality is unclear, but it is thought that the disease primarily affects quality of life, and not lifespan.
Treatment
Standard treatment for ulcerative colitis depends on extent of involvement and disease severity. The goal is to induce remission initially with medications, followed by the administration of maintenance medications to prevent a relapse of the disease. The concept of induction of remission and maintenance of remission is very important. The medications used to induce and maintain a remission somewhat overlap, but the treatments are different. Physicians first direct treatment to inducing a remission which involves relief of symptoms and mucosal healing of the lining of the colon and then longer term treatment to maintain the remission.
Drugs used
Aminosalicylates
Sulfasalazine has been a major agent in the therapy of mild to moderate UC for over 50 years. In 1977 Mastan S. Kalsi et al. determined that 5-aminosalicylic acid (5-ASA and mesalazine) was the therapeutically active compound in sulfasalazine. Since then many 5-ASA compounds have been developed with the aim of maintaining efficacy but reducing the common side effects associated with the sulfapyridine moiety in sulfasalazine.[29]
- Mesalazine, also known as 5-aminosalicylic acid, mesalamine, or 5-ASA. Brand name formulations include Apriso, Asacol, Pentasa, Mezavant, Lialda, and Salofalk.
- Sulfasalazine, also known as Azulfidine.
- Balsalazide, also known as Colazal or Colazide (UK).
- Olsalazine, also known as Dipentum.
Corticosteroids
- Cortisone
- Prednisone
- Prednisolone
- Cortifoam
- Hydrocortisone
- Methylprednisolone
- Beclometasone
- Budesonide - under the brand name of Entocort
Immunosuppressive drugs
- Mercaptopurine, also known as 6-Mercaptopurine, 6-MP and Purinethol.
- Azathioprine, also known as Imuran, Azasan or Azamun, which metabolises to 6-MP.
- Methotrexate, which inhibits folic acid
- Tacrolimus
- Thioguanine, Also known as Lanvis and usually given to cancer patients
Biological treatment
- Infliximab, also known as Remicade.
- Visilizumab
- Adalimumab, also known as HUMIRA.[19]
- Vedolizumab (MLN0002), currently under testing by Millennium Pharmaceuticals[30]
Surgery
Unlike Crohn's disease, ulcerative colitis can generally be cured by surgical removal of the large intestine. This procedure is necessary in the event of: exsanguinating hemorrhage, frank perforation or documented or strongly suspected carcinoma. Surgery is also indicated for patients with severe colitis or toxic megacolon. Patients with symptoms that are disabling and do not respond to drugs may wish to consider whether surgery would improve the quality of life.
Ulcerative colitis (UC) is a disease that affects many parts of the body outside the intestinal tract. In rare cases the extra-intestinal manifestations of the disease may require removal of the colon.[15]
Another surgical option for Ulcerative Colitis that is affecting most of the large bowel, is called the ileo-anal pouch procedure. This procedure is a 2 to 3 step procedure in which the large bowel is removed, except for the rectal stump and anus, and a temporary ileostomy is made. The next part of the surgery can be done in 1 or 2 steps and is usually done at 6 - 12 month intervals from each prior surgery. In the next step of the surgery an internal pouch is made of the patients' own small bowel and this pouch is then hooked back up internally to the rectal stump so that patient can once again have a reasonably functioning bowel system, all internal. The temporary ileostomy can be reversed at this time so that the patient is now internalized for bowel functions, or in another step to the procedure, the pouch and rectal stump anastamosis can be left inside the patient to heal for some time, while the patient still uses the ileostomy for bowel function. Then on a subsequent surgery the ileostomy is reversed and the patient has internalized bowel function again.
Alternative treatments
About 21% of inflammatory bowel disease patients use alternative treatments.[31] A variety of dietary treatments show promise, but they require further research before they can be recommended.[32]
In vitro research, animal evidence, and limited human study suggest that melatonin may be beneficial.[33]
Smoking
Unlike Crohn's disease, ulcerative colitis has a lesser prevalence in smokers than non-smokers. Nonetheless, the risks of smoking contraindicate using cigarettes as a treatment for ulcerative colitis. Patients who choose to use smoking as a treatment should give serious consideration to the links between smoking cessation and the onset or relapse of ulcerative colitis.[34][35]
Dietary modification
Dietary fibre, meaning indigestible plant matter, has been recommended for decades in the maintenance of bowel function. Of peculiar note is fibre from brassica, which seems to contain soluble constituents capable of reversing ulcers along the entire human digestive tract before it is cooked.[36] Oatmeal is also commonly prescribed.
Fats and oils
- Fish oil. Eicosapentaenoic acid (EPA), derived from fish oil. This is an Eicosanoid that inhibits leukotriene activity, the latter which may be a key factor of inflammation. As an IBD therapy, there are no conclusive studies in support and no recommended dosage. But dosages of EPA between 180 to 1500 mg/day are recommended for other conditions, most commonly cardiac.[37]
- Short chain fatty acid (butyrate) enema. The colon utilizes butyrate from the contents of the intestine as an energy source. The amount of butyrate available decreases toward the rectum. Inadequate butyrate levels in the lower intestine have been suggested as a contributing factor for the disease. This might be addressed through butyrate enemas. The results however are not conclusive.
Herbals
- Herbal medications are used by patients with ulcerative colitis. Compounds that contain sulphydryl may have an effect in ulcerative colitis (under a similar hypothesis that the sulpha moiety of sulfasalazine may have activity in addition to the active 5-ASA component).[38] One randomized control trial evaluated the over-the-counter medication methionine-methyl sulphonium chloride (abbreviated MMSC, but more commonly referred to as Vitamin U) and found a significant decreased rate of relapse when the medication was used in conjunction with oral sulfasalazine.[39]
- Boswellia is an Ayurvedic (Indian traditional medicine) herb, used as a natural alternative to drugs. One study has found its effectiveness similar to sulfasalazine.[40]
Bacterial recolonization
- Probiotics may have benefit. One study which looked at a probiotic known as VSL#3 has shown promise for people with ulcerative colitis.[41]
- Fecal bacteriotherapy involves the infusion of human probiotics through fecal enemas.[42] It suggests that the cause of ulcerative colitis may be a previous infection by a still unknown pathogen. This initial infection resolves itself naturally, but somehow causes an imbalance in the colonic bacterial flora, leading to a cycle of inflammation which can be broken by "recolonizing" the colon with bacteria from a healthy bowel. There have been several reported cases of patients who have remained in remission for up to 13 years.[43]
Intestinal parasites
Inflammatory bowel disease is less common in the developing world. Some have suggested that this may be because intestinal parasites are more common in underdeveloped countries. Some parasites are able to reduce the immune response of the intestine, an adaptation that helps the parasite colonize the intestine. The decrease in immune response could reduce or eliminate the inflammatory bowel disease.
Helminthic therapy using the whipworm Trichuris suis has been shown in a randomized control trial from Iowa to show benefit in patients with ulcerative colitis. The therapy tests the hygiene hypothesis which argues that the absence of helminths in the colons of patients in the developed world may lead to inflammation. Both helminthic therapy and fecal bacteriotherapy induce a characteristic Th2 white cell response in the diseased areas, which is somewhat paradoxical given that ulcerative colitis immunology was thought to classically involve Th2 overproduction.[44]
Ongoing research
Recent evidence from the ACT-1 trial suggests that infliximab may have a greater role in inducing and maintaining disease remission.
An increased amount of colonic sulfate-reducing bacteria has been observed in some patients with ulcerative colitis, resulting in higher concentrations of the toxic gas hydrogen sulfide. The role of hydrogen sulfide in pathogenesis is unclear. It has been suggested that the protective benefit of smoking that some patients report is due to hydrogen cyanide from cigarette smoke reacting with hydrogen sulfide to produce the nontoxic isothiocyanate. Another unrelated study suggested sulphur contained in red meats and alcohol may lead to an increased risk of relapse for patients in remission.[16]
There is much research currently being done to elucidate further genetic markers in ulcerative colitis. Linkage with Human Leukocyte Antigen B-27, associated with other autoimmune diseases, has been proposed.
Low dose naltrexone is under study for treatment of Crohn's disease and ulcerative colitis.
See also
- Crohn's disease
- Ileo-anal pouch
- Ileostomy
- Inflammatory bowel disease
- Primary sclerosing cholangitis
- True Guts
- Pyostomatitis vegetans
References
- ↑ 1.0 1.1 Ulcerative colitis at eMedicine
- ↑ Orholm M, Binder V, Sørensen TI, Rasmussen LP, Kyvik KO (2000). "Concordance of inflammatory bowel disease among Danish twins. Results of a nationwide study". Scand. J. Gastroenterol. 35 (10): 1075–81. doi:10.1080/003655200451207. PMID 11099061.
- ↑ Tysk C, Lindberg E, Jarnerot G, Floderus-Myrhed B (1988). ""Ulcerative colitis and Crohn's disease in an unselected population of monozygotic and dizygotic twins. A study of heritability and the influence of smoking". Gut 29 (7): 990–996. doi:10.1136/gut.29.7.990. PMID 3396969.
- ↑ 4.0 4.1 Baumgart DC, Carding SR (2007). ""Inflammatory bowel disease: cause and immunobiology"". The Lancet 369 (9573): 1627–1640. doi:10.1016/S0140-6736(07)60750-8. PMID 17499605.
- ↑ Cho JH, Nicolae DL, Ramos R, et al. (2000). "Linkage and linkage disequilibrium in chromosome band 1p36 in American Chaldeans with inflammatory bowel disease". Hum. Mol. Genet. 9 (9): 1425–32. doi:10.1093/hmg/9.9.1425. PMID 10814724. http://hmg.oxfordjournals.org/cgi/content/full/9/9/1425.
- ↑ Järnerot G, Järnmark I, Nilsson K (1983). "Consumption of refined sugar by patients with Crohn's disease, ulcerative colitis, or irritable bowel syndrome". Scand. J. Gastroenterol. 18 (8): 999–1002. doi:10.3109/00365528309181832. PMID 6673083.
- ↑ Corrao G, Tragnone A, Caprilli R, et al. (1998). "Risk of inflammatory bowel disease attributable to smoking, oral contraception and breastfeeding in Italy: a nationwide case-control study. Cooperative Investigators of the Italian Group for the Study of the Colon and the Rectum (GISC)". Int J Epidemiol 27 (3): 397–404. doi:10.1093/ije/27.3.397. PMID 9698126. http://ije.oxfordjournals.org/cgi/pmidlookup?view=long&pmid=9698126.
- ↑ Voreacos, David (2007-05-29). "Roche Found Liable in First Of 400 Suits Over Accutane". Washington Post. http://www.washingtonpost.com/wp-dyn/content/article/2007/05/29/AR2007052901946.html. Retrieved 2010-05-26.
- ↑ Reddy D, Siegel CA, Sands BE, Kane S (2006). "Possible association between isotretinoin and inflammatory bowel disease". Am. J. Gastroenterol. 101 (7): 1569–73. doi:10.1111/j.1572-0241.2006.00632.x. PMID 16863562.
- ↑ Borobio E, Arín A, Valcayo A, Iñarrairaegui M, Nantes O, Prieto C (2004). "[Isotretinoin and ulcerous colitis]" (in Spanish; Castilian). An Sist Sanit Navar 27 (2): 241–3. PMID 15381956.
- ↑ Reniers DE, Howard JM (2001). "Isotretinoin-induced inflammatory bowel disease in an adolescent". Ann Pharmacother 35 (10): 1214–6. doi:10.1345/aph.10368. PMID 11675849. http://www.theannals.com/cgi/pmidlookup?view=long&pmid=11675849.
- ↑ Heather WonTesoriero (2008-04-23). "Jury Awards $10.5 Million Over Accutane". Wall St Journal. http://online.wsj.com/article/SB120890352155036159.html?mod=dist_smartbrief.
- ↑ Baumgart DC, Sandborn WJ (2007). "Inflammatory bowel disease: clinical aspects and established and evolving therapies.". The Lancet 369 (9573): 1641–57. doi:10.1016/S0140-6736(07)60751-X. PMID 17499606.
- ↑ 14.0 14.1 Fauci et al. Harrison's Internal Medicine, 17th ed. New York: McGraw-Hill Medical, 2008. ISBN 0071599916
- ↑ 15.0 15.1 15.2 15.3 15.4 15.5 15.6 Kornbluth A, Sachar DB (2004). "Ulcerative colitis practice guidelines in adults (update): American College of Gastroenterology, Practice Parameters Committee". Am. J. Gastroenterol. 99 (7): 1371–85. doi:10.1111/j.1572-0241.2004.40036.x. PMID 15233681. http://www.acg.gi.org/physicians/guidelines/UlcerativeColitisUpdate.pdf.
- ↑ 16.0 16.1 Roediger WE, Moore J, Babidge W (1997). "Colonic sulfide in pathogenesis and treatment of ulcerative colitis". Dig. Dis. Sci. 42 (8): 1571–9. doi:10.1023/A:1018851723920. PMID 9286219. http://www.kluweronline.com/art.pdf?issn=0163-2116&volume=42&page=1571.
- ↑ Levine J, Ellis CJ, Furne JK, Springfield J, Levitt MD (1998). "Fecal hydrogen sulfide production in ulcerative colitis". Am. J. Gastroenterol. 93 (1): 83–7. doi:10.1111/j.1572-0241.1998.083_c.x. PMID 9448181.
- ↑ 18.0 18.1 Hanauer SB; Hanauer, Stephen B. (1996). "Inflammatory bowel disease". N. Engl. J. Med. 334 (13): 841–8. doi:10.1056/NEJM199603283341307. PMID 8596552. http://content.nejm.org/cgi/pmidlookup?view=short&pmid=8596552&promo=ONFLNS19.
- ↑ 19.0 19.1 Podolsky DK (2002). "Inflammatory bowel disease". N. Engl. J. Med. 347 (6): 417–29. doi:10.1056/NEJMra020831. PMID 12167685.
- ↑ Shivananda S, Lennard-Jones J, Logan R, et al. (1996). "Incidence of inflammatory bowel disease across Europe: is there a difference between north and south? Results of the European Collaborative Study on Inflammatory Bowel Disease (EC-IBD)". Gut 39 (5): 690–7. doi:10.1136/gut.39.5.690. PMID 9014768. PMC 1383393. http://gut.bmj.com/cgi/pmidlookup?view=long&pmid=9014768.
- ↑ Sonnenberg A, McCarty DJ, Jacobsen SJ (January 1991). "Geographic variation of inflammatory bowel disease within the United States". Gastroenterology 100 (1): 143–9. PMID 1983816.
- ↑ NEJM 2001;344:808.
- ↑ NEJM 1987;316:707.
- ↑ Broomé U, Bergquist A (2006). "Primary sclerosing cholangitis, inflammatory bowel disease, and colon cancer". Semin. Liver Dis. 26 (1): 31–41. doi:10.1055/s-2006-933561. PMID 16496231.
- ↑ Shepherd, NA. (Aug 2002). "Granulomas in the diagnosis of intestinal Crohn's disease: a myth exploded?". Histopathology 41 (2): 166–8. doi:10.1046/j.1365-2559.2002.01441.x. PMID 12147095.
- ↑ Mahadeva, U.; Martin, JP.; Patel, NK.; Price, AB. (Jul 2002). "Granulomatous ulcerative colitis: a re-appraisal of the mucosal granuloma in the distinction of Crohn's disease from ulcerative colitis.". Histopathology 41 (1): 50–5. doi:10.1046/j.1365-2559.2002.01416.x. PMID 12121237.
- ↑ Leighton JA, Shen B, Baron TH, et al. (2006). "ASGE guideline: endoscopy in the diagnosis and treatment of inflammatory bowel disease". Gastrointest. Endosc. 63 (4): 558–65. doi:10.1016/j.gie.2006.02.005. PMID 16564852.
- ↑ Olsson R, Danielsson A, Järnerot G, et al. (1991). "Prevalence of primary sclerosing cholangitis in patients with ulcerative colitis". Gastroenterology 100 (5 Pt 1): 1319–23. PMID 2013375.
- ↑ S. Kane (2006). "Asacol - A Review Focusing on Ulcerative Colitis". http://www.touchalimentarydisease.com/articles.cfm?article_id=6364&level=2.
- ↑ "Millennium Resumes Clinical Program For MLN0002". 18 May 2007. http://www.medicalnewstoday.com/articles/71328.php. Retrieved 5 February 2010.
- ↑ Bensoussan M, Jovenin N, Garcia B, et al. (January 2006). "Complementary and alternative medicine use by patients with inflammatory bowel disease: results from a postal survey". Gastroenterol. Clin. Biol. 30 (1): 14–23. doi:10.1016/S0399-8320(06)73072-X. PMID 16514377. http://www.masson.fr/masson/MDOI-GCB-01-2006-30-1-0399-8320-101019-200517348.
- ↑ Shah S (2007). "Dietary factors in the modulation of inflammatory bowel disease activity". MedGenMed 9 (1): 60. PMID 17435660. PMC 1925010. http://www.medscape.com/viewarticle/553039.
- ↑ Terry PD, Villinger F, Bubenik GA, Sitaraman SV (January 2009). "Melatonin and ulcerative colitis: evidence, biological mechanisms, and future research". Inflamm. Bowel Dis. 15 (1): 134–40. doi:10.1002/ibd.20527. PMID 18626968.
- ↑ Calkins BM (1989). "A meta-analysis of the role of smoking in inflammatory bowel disease". Dig. Dis. Sci. 34 (12): 1841–54. doi:10.1007/BF01536701. PMID 2598752.
- ↑ Lakatos PL, Szamosi T, Lakatos L (2007). "Smoking in inflammatory bowel diseases: good, bad or ugly?". World J Gastroenterol. 13 (46): 6134–9. doi:10.3748/wjg.13.6134. PMID 18069751.
- ↑ Akhtar MS, Munir M (1989). "Evaluation of the gastric anti-ulcerogenic effects of Solanum nigrum, Brassica oleracea and Ocimum basilicum in rats.". Journal of ethnopharmacology 27 (1-2): 163–76. doi:10.1016/0378-8741(89)90088-3. PMID 2515396. http://www.ncbi.nlm.nih.gov/sites/entrez?Db=pubmed&Cmd=DetailsSearch&Term=((%22brassica%22%5BTIAB%5D+NOT+Medline%5BSB%5D)+OR+%22brassica%22%5BMeSH+Terms%5D+OR+cabbage%5BText+Word%5D)+AND+(%22ulcer%22%5BMeSH+Terms%5D+OR+ulcer%5BText+Word%5D). "Brassica oleracea (leaf) powder did not affect the ulcer index significantly but its aqueous extract lowered the index and increased hexosamine levels, suggesting gastric mucosal protection.".
- ↑ "Omega-3 fatty acids, fish oil, alpha-linolenic acid". MedlinePlus Herbs and Supplements. http://www.nlm.nih.gov/medlineplus/druginfo/natural/patient-fishoil.html.
- ↑ Brzezinski A, Rankin G, Seidner D, Lashner B (1995). "Use of old and new oral 5-aminosalicylic acid formulations in inflammatory bowel disease.". Cleve Clin J Med 62 (5): 317–23. PMID 7586488.
- ↑ Salim A (1992). "Role of sulphydryl-containing agents in the management of recurrent attacks of ulcerative colitis. A new approach.". Pharmacology 45 (6): 307–18. doi:10.1159/000139016. PMID 1362613.
- ↑ PMID 9049593 (PubMed)
Citation will be completed automatically in a few minutes. Jump the queue or expand by hand - ↑ Bibiloni R, Fedorak RN, Tannock GW, et al. (2005). "VSL#3 probiotic-mixture induces remission in patients with active ulcerative colitis". Am. J. Gastroenterol. 100 (7): 1539–46. doi:10.1111/j.1572-0241.2005.41794.x. PMID 15984978. VSL#3 company site
- ↑ Borody TJ, Warren EF, Leis SM, Surace R, Ashman O, Siarakas S (2004). "Bacteriotherapy using fecal flora: toying with human motions". J. Clin. Gastroenterol. 38 (6): 475–83. doi:10.1097/01.mcg.0000128988.13808.dc. PMID 15220681. http://meta.wkhealth.com/pt/pt-core/template-journal/lwwgateway/media/landingpage.htm?issn=0192-0790&volume=38&issue=6&spage=475. Fulltext(PDF)
- ↑ Borody TJ, Warren EF, Leis S, Surace R, Ashman O (2003). "Treatment of ulcerative colitis using fecal bacteriotherapy". J. Clin. Gastroenterol. 37 (1): 42–7. doi:10.1097/00004836-200307000-00012. PMID 12811208. http://meta.wkhealth.com/pt/pt-core/template-journal/lwwgateway/media/landingpage.htm?issn=0192-0790&volume=37&issue=1&spage=42. Fulltext(PDF)
- ↑ Summers RW, Elliott DE, Urban JF, Thompson RA, Weinstock JV (2005). "Trichuris suis therapy for active ulcerative colitis: a randomized controlled trial". Gastroenterology 128 (4): 825–32. doi:10.1053/j.gastro.2005.01.005. PMID 15825065. http://linkinghub.elsevier.com/retrieve/pii/S0016508505000259.
External links
- Ulcerative colitis at the Open Directory Project
- Ulcerative Colitis Library: at Health.com
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||